Welcome to Bioresonance according to Paul Schmidt
Rayonex Biomedical represents the cause-oriented treatment approach of Bioresonance according to Paul Schmidt – named after the company founder and engineer Paul Schmidt.
Only a few people were able to change the world like Paul Schmidt did.
His ingenious ideas, which resulted in the granting of more than 300 commercial patents, bear witness to his imagination and creativity.
Contact us for consultation
Get in touch with us and let us convince you of Bioresonance according to Paul Schmidt. We will respond to your questions as thoroughly as possible and provide you with information on how best to use our products for you. We look forward to finding you in one or more of the numerous areas of application.
Bioresonance according to Paul Schmidt explained
The treatment is based on one of nature’s principle: Sunlight is the most well-known form of bioresonance. When sunlight comes in contact with our skin, our skin darkens. This is caused by the UV component of sunlight, which has a frequency spectrum in the high terahertz range.
In 1975 the engineer Paul Schmidt discovered that not only the sun’s frequency spectrum, but also other frequency spectra have an effect on the body.
In short, Bioresonance according to Paul Schmidt is based on stimulating the organism with the aim of supporting self-regulation. It is cause-oriented, universally applicable and has been tried and tested in over 45 countries worldwide.
Rayonex Biomedical GmbH actively researches frequency spectra that are transmitted to the patient during treatment with modern Bioresonance devices – a safe and gentle therapy option, confirmed by clinical studies.
Knowledge transfer takes place regularly in our seminars and webinars. Another source of information is the extensive literature, which among other things with the book “Bioresonance according to Paul Schmidt” offers a standard work, which is now in its sixth edition.
The unique combination of German engineering and an understanding of alternative health care is what makes our medical devices so special.

Wide range of uses
Healthcare professional
Healthcare professional
TRIAL-PROVEN APPLICATIONS OF BIORESONANCE ACCORDING TO PAUL SCHMIDT
Applications based on randomized, prospective, double-blind clinical trials. Please click on the elements below to open or close them.
- "The Rayocomp PS 1000 polar 4.0 bioresonance device achieved significant improvements in all objective and subjective parameters compared to the placebo device."
- "There was no change in the NDI (Neck-Disability-Index) of the placebo group, but a significant improvement in the NDI of the group treated with the Rayocomp bioresonance device."
- "The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- "In terms of physical capacity, patients receiving the placebo treatment showed no significant improvement, while patients treated with the Rayocomp bioresonance device showed significant improvements in all parameters."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
EXPERIENCE-BASED APPLICATIONS OF BIORESONANCE ACCORDING TO PAUL SCHMIDT
Applications based on experience – yet to be proven with randomized, prospective, double-blind clinical trials. Please click on the elements below to open or close them.
"The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
"The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
- Aluminium
- Formaldehyde
- Thiomersal
- Mercury
- Phenols
- Protein carriers
"The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
"The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
"The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
- "The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- "In terms of physical capacity, patients receiving the placebo treatment showed no significant improvement, while patients treated with the Rayocomp bioresonance device showed significant improvements in all parameters."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
- "The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- "In terms of physical capacity, patients receiving the placebo treatment showed no significant improvement, while patients treated with the Rayocomp bioresonance device showed significant improvements in all parameters."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
"The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
"The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
- "The SF-36 parameters of physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social functioning, emotional role functioning and mental health achieved no significant improvement in patients receiving the placebo treatment. Whereas, all of the parameters showed significant improvements in patients receiving treatment with the Rayocomp bioresonance device."
- "In terms of physical capacity, patients receiving the placebo treatment showed no significant improvement, while patients treated with the Rayocomp bioresonance device showed significant improvements in all parameters."
- From the final clinical report by Dr. Hans Werner Voss, published in the book "Successful Treatment of Chronic Pain" by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
Does bioresonance according to Paul Schmidt have Side Effects?
The clinical prospective, double-blind, randomised trial shows that Bioresonance according to Paul Schmidt can be a useful therapy for both the mental and physical effects of chronic pain, with no side-effects:
“No undesirable side effects, contraindications or risks were identified.“
– From the final clinical report by Dr. Hans Werner Voss, published in the book “Successful Treatment of Chronic Pain” by Rayonex Biomedical GmbH from 2020, published in the German Register of Clinical Studies (BfArM).
The trial “Efficacy Study according to system of devices of Rayonex Biomedical” from the renowned Fraunhofer Institute confirms , that Bioresonance according to Paul Schmidt and the RAH meet the requirements of an effective therapy with a low level of side-effects.
Frequently asked questions (FAQ)
Please click on the elements below to open or close them.
- The Rayocomp PS 1000 polar 4.0 is our high-end bioresonance device. The client capability, a large intuitive touch screen and the particularly powerful and fast dipole antenna system are special features. The Rayoscan® is the high-end software module exclusively for the Rayocomp PS 1000 polar 4.0. Through a fully automatic, energetic test based on electrocardiogram (ECG) analysis, the therapist is presented with a fully comprehensive, cause-oriented therapy suggestion as a result.
- Despite its small size, our Rayocomp PS 10 has it all - an integrated powerful battery, the modular design and the highest industry standards; a device lighter and more compact than most laptops.
Studies & scientific works

Randomized, prospective, double-blind clinical trial in patients with cervical spine syndrome.

Bioresonance therapy according to Paul Schmidt - Does it work?
Scientifically proven!

The basis of all our developments is Bioresonance approach according to Paul Schmidt.
Rayonex Biomedical GmbH has conducted several studies on Bioresonance according to Paul Schmidt over the last few years. The most meaningful of which is the new clinical prospective, double-blind, randomised study on pain reduction in patients with cervical spine syndrome. This study proves that, in the truest sense, Bioresonance according to Paul Schmidt works based on the idea that success comes in waves.
Thus, the results of the study confirm what users have been saying for decades:
Bioresonance therapy according to Paul Schmidt relieves symptoms and helps give patients a new sense of physical well-being.
High recommendation rates
„Overall, there were significant differences in how patients answered the question of whether they would recommend the treatment to others. All patients in the Rayocomp group said that they would recommend it, while only around a quarter of patients in the placebo group said the same.“
ASSESSMENT OF THE THERAPY |
CONTROL GROUP (PLACEBO) |
EXPERIMENTAL GROUP (RAYOCOMP) |
||
|---|---|---|---|---|
| Number (n) | Proportion % | Number (n) | Proportion % | |
| Therapy recommendation | 6 | 26,1% | 24 | 100,0% |
| No recommendation of the therapy | 17 | 73,9% | 0 | 0,0% |
FROM THE FINAL CLINICAL REPORT BY DR. HANS WERNER VOSS, PUBLISHED IN THE BOOK “SUCCESSFUL TREATMENT OF CHRONIC PAIN” BY RAYONEX BIOMEDICAL GMBH FROM THE YEAR 2020.
Bioresonance according to Paul Schmidt - does it work?
Proven Efficacy
During the course of the development, Bioresonance according to Paul Schmidt has been able to demonstrate its capabilities time and again in both in-vitro and in-vivo studies.
For example, cell biological studies (i.a. at the Fraunhofer Institute) showed that the cell activity under the influence of Bioresonance according to Paul Schmidt could be increased by up to 45%. In-vivo studies have also been conducted into a wide range of topics.
Rayonex Biomedical has conducted several studies on Bioresonance according to Paul Schmidt over the last few years. The most meaningful of which is the new clinical prospective, double-blind, randomised study on pain reduction in patients with cervical spine syndrome. This study proves that, in the truest sense, Bioresonance according to Paul Schmidt works based on the idea that success comes in waves.
Bioresonance therapy device Rayocomp PS 1000 polar 4.0
Our flagship for human and veterinarian application
By means of our selection of diverse, suitable accessory products.
If you wish, you can also receive a certification that you can also show to your clients.
An intuitively understandable touchscreen display for therapy and work with the client.
Use the 360 degree animation. Rotate the image by clicking on it and moving the mouse left and right.
Multiple profiles can be set up.
For short-term protection in the event of a power failure.
Internet software updates, highest possible and robust quality of all device components used.
Bioresonance therapy device Rayocomp PS 10
Our mobile device for human and veterinarian usage
For uninterrupted therapy and use.
Intuitive operability.
Maximum customisation and low cost.
Use the 360 degree animation. Rotate the image by clicking on it and moving the mouse left and right.
No extensive training in the device necessary.
Highest possible and robust quality of all device components used
Rayocomp PS 10 - perfect for home therapy
Explanation of the Rayocomp PS 10 for home therapy. Start the video by clicking on the preview image!
The Rayoscan® in the veterinary sector
Explanation of the Rayoscan for home therapy. Start the video by clicking on the preview image!
The easy handling speaks for itself!
Please click on the switcher below to switch between the two options.
PS 10 Human
PS 10 VET
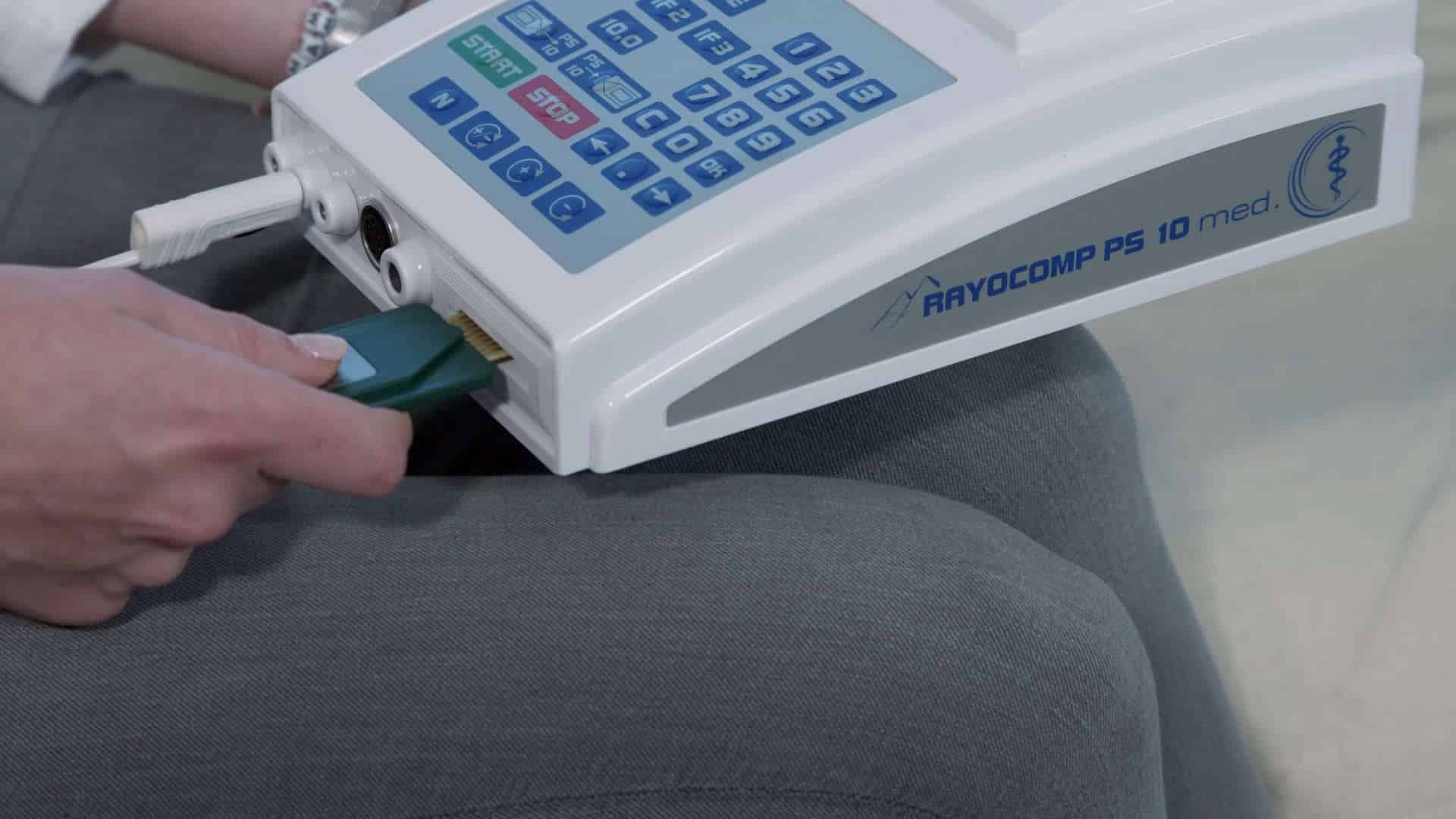
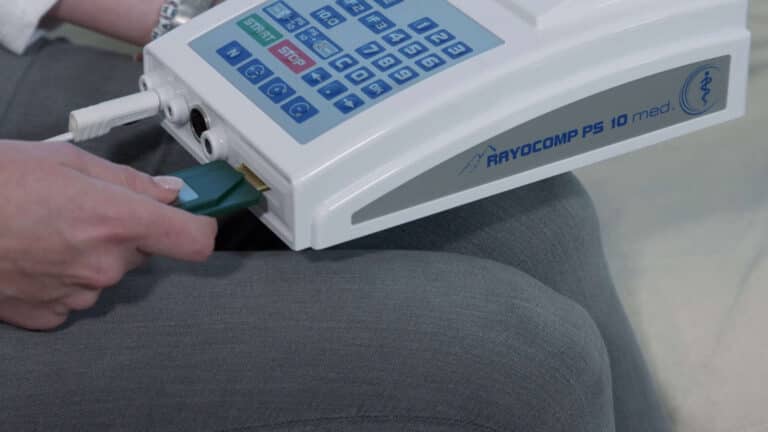
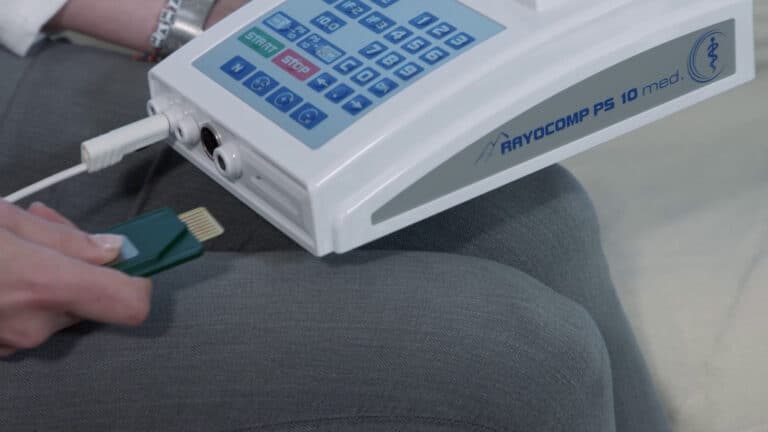
Switch on the device.
Connect the area detector.
Insert the Green-Card, wait briefly.
Remove the Green-Card.
Let's go!



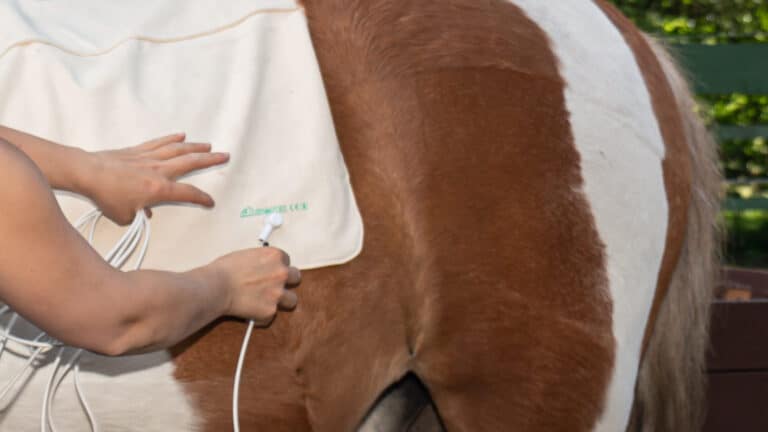
Putting on the area detector.
Switch on the device.
Connect the area detector.
Let's go!



THE TYPICAL TREATMENT PROCEDURE
Please click on the switcher below to switch between the two options.
Analysis & Testing
Hometherapy
Bioresonance according to Paul Schmidt - Experiences, Testimonials & Reviews
Please click on the side arrows or dots below the testimonials to navigate.

Please note that the customer testimonials listed here are based on the personal experiences of our customers and represent their own opinions. The information, advice and solution approaches presented cannot replace a visit to an alternative practitioner or a naturopathic doctor. Furthermore, we would like to point out that classical conventional medicine has neither accepted nor recognized the effect of bioenergetic oscillations.
More from our product world of Bioresonance according to Paul Schmidt

Biofield-forming
For location-specific influences such as 5G networks, WLAN, screen radiation and more.

The Rayonex nutrient concept
The holistic supplement concept for human and veterinarian nutrition - based on the principles of Bioresonance according to Paul Schmidt.
WHAT SETS US APART
-
Personal support
Global network of partners.
-
Medical device company
Established in 1982.
-
Certified
According to DIN EN ISO 13485: 2016 for the development, production and sale of medical devices.
-
Made in Germany
CO2-neutral and high-precision with robot support in our own manufactory.
-
Therapy proven worldwide
Present in 46 countries around the world.
-
Universal
Application to humans and animals.
-
Cause-oriented
Treatment approach of Bioresonance according to Paul Schmidt.
-
Sauerland Pyramids
Our company buildings are as extraordinary as our products.
Who was Paul Schmidt?
Paul Schmidt was born in the Sauerland area of Germany in 1922. Due to his father’s early death, his childhood was marked by deprivation and the need to improvise. Early on he developed great technical skills, which led to an education in engineering science. As an engineer, he busied himself with the development of agricultural machines, before founding his own enterprise, the Tracto-Technik company. While building up this company he continued his commitment to social and cultural affairs for which he was awarded the Bundesverdienstkreuz (The Federal Cross of Merit of Germany).
It was his social awareness which led him to apply his engineering expertise to the field of alternative medicine, a work which resulted in today’s Bioresonance according to Paul Schmidt. In 1982 he founded the Rayonex Biomedical GmbH company, in order to develop his concepts of alternative medicine. Until his death on the 2nd of September 1994, Paul Schmidt continued his social commitment. He sketched children’s books, which he provided to kindergartens and schools at no charge.
Manufactory in Germany
In terms of origin, we are locally rooted: Rayonex Biomedical develops all products Made in Germany in Lennestadt in the distinctive Sauerland pyramids, the company’s headquarters.
Manufacturing takes place in the neighbouring Rayonex manufactory – CO2-neutral and highly precise with robot support. Rayonex Biomedical is certified according to DIN EN ISO 13485:2016 for the development, production and distribution of medical products and is audited annually by TÜV Nord.
Our philosophy
Rayonex Biomedical has set itself the goal of improving the quality of life and well-being of humans and animals worldwide through the positive effects of Bioresonance according to Paul Schmidt, while at the same time creating a new awareness of the effect of frequencies on the organism.
Is it safe? Our Product certifications
Effective proof that a product meets all relevant safety and quality requirements and has the respective required product features – the certifications awarded to us are visible proof for you and serve as a reliable decision-making aid, since in this process we as Rayonex Biomedical and our products are tested by highest recognized independent third parties.
Please click on the logos above to download/view the certficates.
Worldwide network of partners
Rayonex Biomedical, based in Lennestadt (Germany), is an established medical products company since 1982 and currently represented in 46 countries around the world.
If you are interested in our products and services or becoming a partner, we are delighted to get in touch with you!
THE COLLECTED KNOWLEDGE IN ONE STANDARD WORK
Treatment does not have to be the first step to recovery!
Rather, it is up to us to take the first step towards healing our bodies and staying healthy by eliminating interference fields.
But which holistic test can we apply to find out whether a health burden is caused by electrosmog, geopathic stress zones, bacteria, viruses or parasites?
What about our acid-base balance? Do we have any nutritional deficiencies of minerals, trace elements or other major dietary components?
How can we find out if our primary staple, our water, really meets the quality requirements of our body? As early as 1976, through his discoveries, Paul Schmidt laid the foundation for answering these questions.
The author Dipl-Ing. (TH) Dietmar Heimes, managing director of Rayonex Biomedical GmbH, has been developing and designing Bioresonance according to Paul Schmidt since 1994.
The book is aimed at health care practitioners and their patients, as well as individuals who love the holistic and gentle approaches of alternative medicine.

International Webinars
Please note that the given information, advices and solution approaches are originated from more than 40 years of experience. But they can not replace the consultation of a medical practitioner or a naturopathy orientated doctor.